All articles by Blatha
Sourcing on a global scale
As more clinical trials are conducted across different regions, it is important for the industry to address the problems in the clinical supply chain that could affect the drug development process as a whole. Dr Cristina Chang, vice-president global clinical development and medical affairs at OBI Pharma, examines the current challenges, as well as critical success factors in clinical trial materials supply for comparator sourcing, and outlines how to simplify the process, eliminate redundancy, and stay ahead of the competition through enhancing customer satisfaction and efficiency.
Sweet harmony: clinical trials protocol template
The increasing complexity of clinical trials, combined with the inexperience of many investigators who write the protocol templates, has long caused concerns. In 2017, US officials responded to the lack of consistency across clinical trial protocols by creating their own model template for investigators to use. How well has it gone down? Neil Thompson explores.
Dispersion: advancing drug delivery
New developments in the formulation of amorphous solid dispersions are an important facet of advancing new drug delivery systems. World Pharmaceuticals Frontiers explores recent developments in this area, focusing on its four major market offerings.
The power of cannabinoids
Though medical marijuana has been available throughout the Western world for years, researchers are now examining the possibilities of using cannabis-based drugs to fight a range of diseases, including cancers and epilepsy. Andrea Valentino talks to Dr Ingo Michels, former head of office at the German Federal Drug Commission, about how the industry is advancing, and where it might go in future.
Forward together: CDMOs
Even small innovations from service providers can lead to big outcomes in pharma, but aligning motivations across the supply chain is a must. Dr Uwe Bücheler, Boehringer Ingelheim’s corporate senior vice-president, tells Kim Thomas how to ensure that all parties are working towards the same goals.
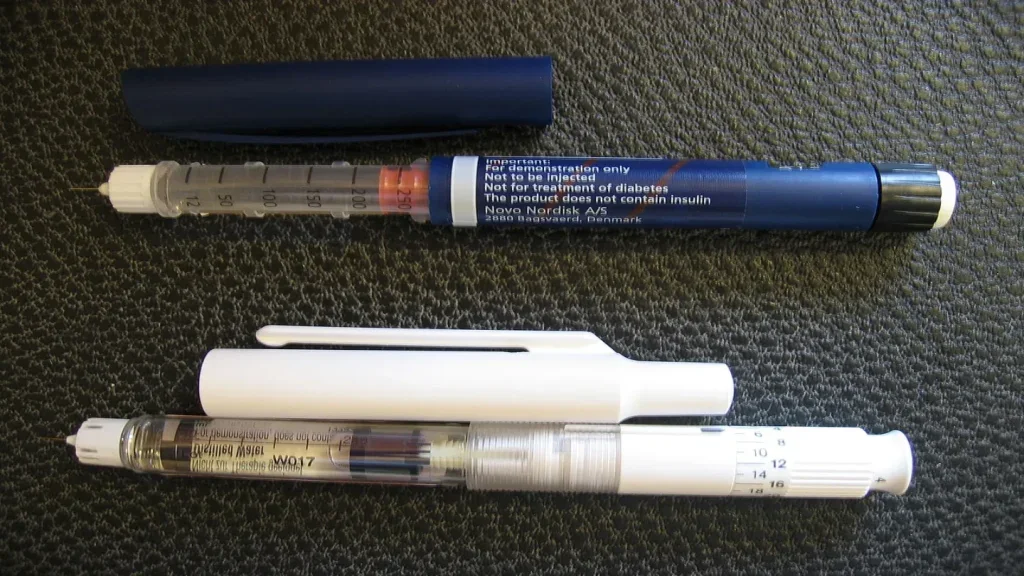
Needles: soon to be obsolete?
Needle manufacturers are embracing new polymers while an ageing population is driving the demand for safe prefilled syringes – but could pharma be on the verge of getting rid of needles entirely? Eleanor Wilson explores syringe safety innovations and new developments in needle-free technology.
The changing face of oral medicines
For the delivery of vaccines and allergens, the buccal and sublingual routes have been extensively investigated. Consequently, good knowledge of the immune cells distributed within the oral mucosal tissue has become increasingly important. World Pharmaceutical Frontiers highlights the current status of this drug delivery method, and explores the possibilities and limitations with regard to quality, safety and efficacy issues.
Out cold: developments in pharma cold chain
When transporting a drug or biological sample, it’s essential to maintain proper temperature control, with many of the newer drugs on the market requiring cold chain handling. With the plethora of packaging options available – how can pharma companies be sure they are making the right choices? Jorg Grigo, director of Grünenthal, talks with Abi Millar about the importance of packaging and how these new developments are changing the face of the pharma cold chain.
Securing the supply chain
David Brown, vice-president of technical operations at Alcresta Pharmaceuticals, talks to World Pharmaceutical Frontiers about the importance of securing reliable partners within the entire continuum of a project’s development, and the need to constantly evaluate your supplier network.
The indefinite particle
Jonas Hoeg Thygesen, research scientist at the Microanalysis Centre, Novo Nordisk, speaks to Kerry Taylor-Smith about methods and techniques for particle analysis and characterisation. He discusses the strengths and weaknesses of current analytical methods for particle identification and characterisation, and examines future methods.