All articles by Vidya Sagar Maddela
Vivace Therapeutics secures FDA orphan drug status for VT3989
VT3989 is an investigational small-molecule cancer therapeutic designed to target the Hippo pathway by inhibiting palmitoylation of TEAD protein family members.
J&J seeks FDA approval for new Tremfya label evidence
The application seeks to include new evidence showing significant inhibition of joint structural damage in adults with active psoriatic arthritis.
ViroCell, AvenCell collaborate on retroviral vector for CAR-T therapies
The partnership aims to accelerate the development of AVC-203, a CD19/CD20 dual-targeted cell therapy designed to treat B-cell malignancies and autoimmune diseases.
Boehringer Ingelheim, Re-Vana collaborate on ophthalmic therapies
Boehringer Ingelheim and Re-Vana aim to create extended-release treatments for eye diseases using Re-Vana’s drug delivery technology, advancing ocular therapeutics.
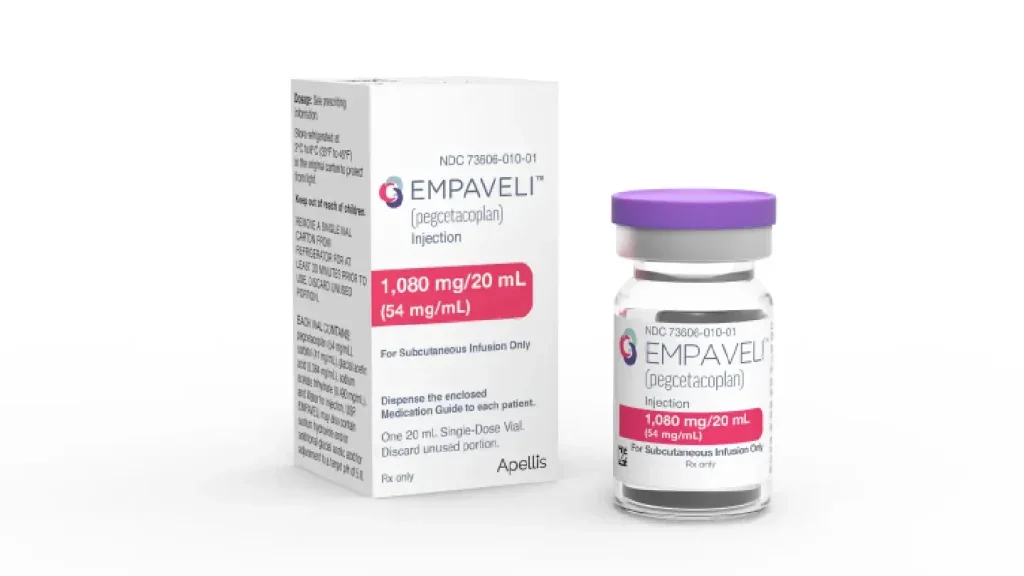
Apellis secures FDA approval for Empaveli to treat rare kidney diseases
Empaveli is a targeted C3 therapy designed to regulate the excessive activation of the complement cascade, which is linked to serious diseases progression.
Hengrui Pharma, GSK enter licensing deal worth up to $12bn
GSK will obtain a global license to Hengrui Pharma’s potential PDE3/4 programme, HRS-9821, except in mainland China, Hong Kong, Macau and Taiwan.
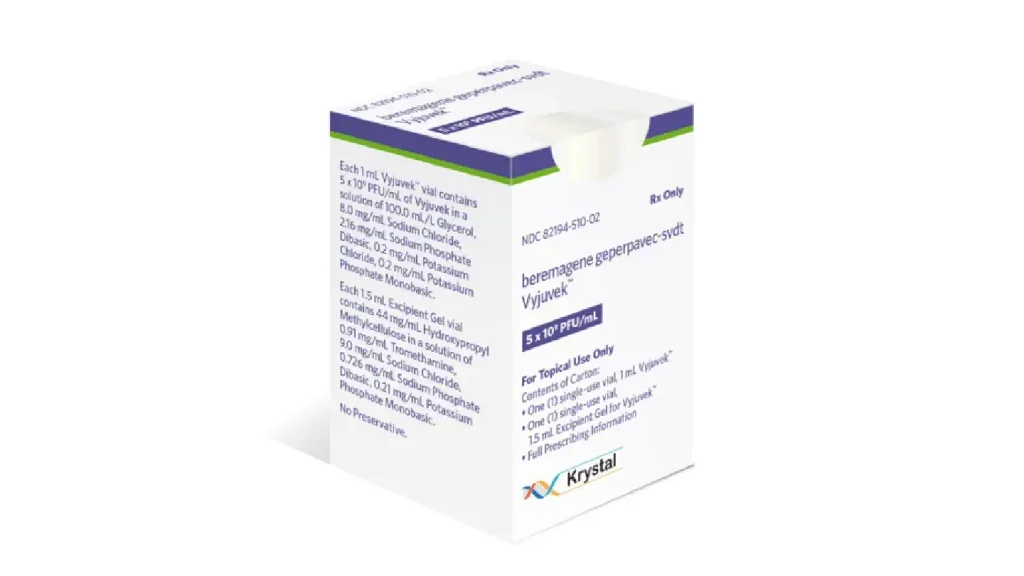
Krystal Biotech’s Vyjuvek authorised in Japan to treat DEB
Vyjuvek, which targets the root cause of DEB by delivering functional copies of the COL7A1 gene, can be administered at home or in healthcare settings.
Ipsen secures EC approval for Cabometyx in neuroendocrine tumours
Cabometyx was originally developed by Exelixis, a biopharmaceutical company focused on oncology, and Ipsen secured exclusive rights to the drug outside the US and Japan.
Matchpoint, Novartis partner for inflammatory disease treatments
Matchpoint is eligible to receive up to $1bn in total payments, including an upfront payment of $60m and research funding, milestone payments, and tiered royalties.
US FDA extends review period for GSK’s Blenrep in multiple myeloma
Blenrep combinations are intended for the treatment of patients with relapsed or refractory multiple myeloma who have received at least one prior line of therapy.